1. Which patients with neutropenic fever do you consider for outpatient management?
Neutropenic fever is a common presentation to the Emergency Department, especially in tertiary hospitals where many oncology patients are undergoing chemotherapy. According to the Infectious Disease Society of America (IDSA), fever in neutropenic patients is defined as a single oral temperature of >38.3°C (101°F) or a temperature of >38.0°C (100.4°F) sustained for >1 hour. Rectal temperature measurements (and rectal exams) are not recommended by the IDSA to prevent colonizing gut organisms from entering the surrounding mucosa and soft tissues (Freifeld, 2011). The definition of neutropenia varies from institution to institution; the IDSA defines it as an (ANC) <500 cells/microL or an ANC that is expected to decrease to <500 cells/microL over the next 48 hours. Profound or severe neutropenia occurs when the ANC is < 100 cells/micromol. The National Cancer Institute defines neutropenia as an ANC < 1000 cells/micromol (HHS 2010).
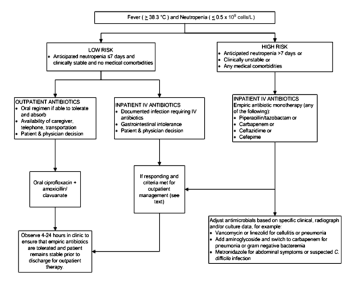
Patients with neutropenic fever are usually started on broad spectrum IV antibiotics and admitted to the hospital, however there are a subgroup of patients who can be safely managed as outpatients. The official wording from the IDSA guidelines is that “Carefully selected low-risk patients may be candidates for oral and/or outpatient empirical antibiotic therapy (B-I)”. Grade B is defined as moderate evidence to support a recommendation for use, and Level I is evidence from ≥1 properly randomized, controlled trial. The data which they derived these recommendations include one large series of patients, where oral outpatient treatment for low-risk fever and neutropenia was deemed successful in 80% of patients, with 20% patient requiring readmission. Factors predicting readmission include age > 70, grade of mucositis >2, poor performance status, and ANC < 100 cells/microL at onset of fever (Escalante 2006). Klastersky et al studied 178 low-risk patients who were treated with oral antibiotics. Only 3 patients were readmitted resulting in a 96% success rate (Klastersky 2006) .
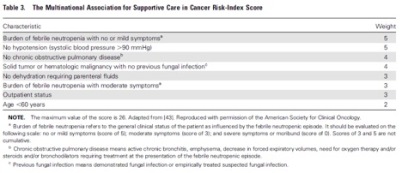
The IDSA formally risks stratifies using the Multinational Association for Supportive Care in Cancer (MASCC) scoring system. The adult guidelines from Australia, European Society for Medical Oncology (ESMO), the American Society of Clinical Oncology (ASCO) also recommend the use of the MASCC index (Gea-Banacloche 2013). Low-risk patients have a MASCC score ≥ 21.
The index has been validated in multiple settings and performs well, although it may function better in solid tumors than in hematologic malignancies (Klastersky 2013). An issue with the major criteria of “burden of febrile neutropenia” is that there is no standardized definition for this criteria, making uniform application of the MASCC confusing (Kern 2006).
Based on the best available data prior to the 2010 guidelines release, the IDSA developed an algorithm to risk stratify neutropenic fever patient and their appropriate management:
Ciprofloxacin plus amoxicillin-clavulanate is recommended for oral empirical treatment (Friefeld 2010). Other oral regimens, including levofloxacin or ciprofloxacin monotherapy or ciprofloxacin plus clindamycin, are less well studied but are commonly used. In low-risk patients, the risk of invasive fungal infection is low, and therefore routine use of empirical antifungal therapy is not recommended. Respiratory virus testing and chest radiography are indicated for patients with upper respiratory symptoms and/or cough .
A systematic review and meta-analysis of 14 RCTs was published in 2011 and not, therefore, included in the 2010 guidelines (Teuffel 2011). The meta-analysis concluded that inpatient versus outpatient management was not significantly associated with treatment failure; death occurred with no difference between the two groups; and outpatient oral versus outpatient parenteral antibiotics were similarly efficacious with no association between route of administration and treatment failure.
It must be said without questions that the patients whom you are treating as outpatients need to have easily accessible and very close follow up with their oncologists. They should be vigilantly examined for a source of infection, including a thorough skin, mucosal, and neurologic exam, as any obvious focal infection may necessitate inpatient treatment.
Bottom Line: Based on the IDSA guidelines and other meta-analyses, it is reasonable to treat a certain subset of low risk febrile neutropenic patients with oral antibiotics as an outpatient with good follow up.
2. Which patients with community-acquired pneumonia do you admit?
Community-acquired pneumonia (CAP) is defined as an acute infection of the pulmonary parenchyma in a patient who has acquired the infection in the community, as distinguished from hospital-acquired (nosocomial) pneumonia, which occurs 48 hours or more after hospital admission and is not present at time of admission. A third category of pneumonia, designated “healthcare-associated pneumonia,” is acquired in other healthcare facilities such as nursing homes, dialysis centers, and outpatient clinics or within 90 days of discharge from an acute or chronic care facility. The most common validated prediction rules for prognosis in community-acquired pneumonia include the Pneumonia Severity Index, CURB, and CURB-65 severity scores.
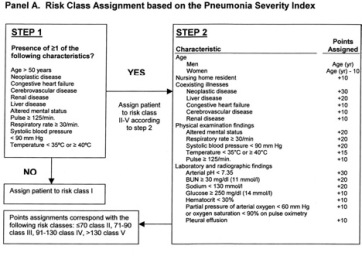
According to the Pneumonia Severity Index (PSI), patients in risk classes I-III are define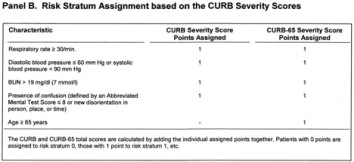 d as low risk for short-term mortality and are considered for outpatient treatment. In the original derivation study of the PSI, Mortality ranged from 0.1 to 0.4 percent for class I patients, from 0.6 to 0.7 percent for class II, and from 0.9 to 2.8 percent for class III (Fine 1997). This older but well validated rule can be difficult to remember and apply; as a result, a relatively simpler CURB score with a total point score ranging from 0-4 was described and externally validated (Ewig 2004). A modified version, the CURB-65, which added age ≥ 65 as another positive risk factor was internally validated to stratify short term mortality for patients with CAP (Lim 2003). Patients scoring CURB < 1 and CURB-65 < 2 are considered low-risk and candidates for outpatient treatment. The CURB and CURB-65 scores are easier to remember and apply, however it does require laboratory data (BUN), whereas the lowest PSI risk class I can be attained without getting blood draws, a benefit appreciated in outpatient settings.
d as low risk for short-term mortality and are considered for outpatient treatment. In the original derivation study of the PSI, Mortality ranged from 0.1 to 0.4 percent for class I patients, from 0.6 to 0.7 percent for class II, and from 0.9 to 2.8 percent for class III (Fine 1997). This older but well validated rule can be difficult to remember and apply; as a result, a relatively simpler CURB score with a total point score ranging from 0-4 was described and externally validated (Ewig 2004). A modified version, the CURB-65, which added age ≥ 65 as another positive risk factor was internally validated to stratify short term mortality for patients with CAP (Lim 2003). Patients scoring CURB < 1 and CURB-65 < 2 are considered low-risk and candidates for outpatient treatment. The CURB and CURB-65 scores are easier to remember and apply, however it does require laboratory data (BUN), whereas the lowest PSI risk class I can be attained without getting blood draws, a benefit appreciated in outpatient settings.
The three prediction rules were pitted against each other in a prospective study of over 3000 patients with community-acquired pneumonia from 32 hospital EDs to see which one was better at predicting 30-day mortality (Aujesky, 2005). Inclusion criteria included age ≥ 18 with a clinical diagnosis of pneumonia and new radiographic pulmonary infiltrate. Exclusion criteria included hospital acquired pneumonia, immunosuppression, psychosocial problems incompatible with outpatient treatment, or pregnancy.
The PSI classified a significantly greater proportion of patients as low risk (68%) than the CURB (51%) and the CURB-65 (61%). Of the low-risk by PSI (class I-III), the aggregate 30-day mortality was 1.4%, which is less than the CURB (score < 1) low-risk mortality rate of 1.7% and also 1.7% mortality for CURB-65 low-risk group (score <2). High-risk patients based on the PSI (class IV-V) had a higher mortality of 11.1% compared with high-risk CURB (≥1) and high risk CURB-65 (≥2), with respective mortality rates of 7.6% and 9.1%.
The PSI had a slightly higher sensitivity and negative predictive value across each risk cut-off point compared to the CURB and CURB-65. In addition, by comparing the areas under the receiver operating characteristic (ROC) curves, the PSI had a statistically significantly greater discriminatory power to predict 30-day mortality. CURB-65 showed a higher overall discriminatory power than the original CURB score.
Based on the estimation of 4 million annual cases of community acquired pneumonia in the USA (DeFrances, 2007), and the average cost of inpatient versus outpatient care of $7500 vs $264; using the PSI would identify an additional 650,000 low-risk patients vs. CURB and 250,000 vs. CURB-65, saving a significant amount of healthcare dollars, while still maintaining a low 30-day mortality rate (Aujesky, 2005) .
The 2007 (most current) Infectious Diseases Society of America (IDSA) recommendations on managing CAP revolve around the initial assessment of severity. “Severity-of-illness scores such as the CURB-65 criteria, or prognostic models, such as the Pneumonia Severity Index (PSI), can be used to identify patients with CAP who may be candidates for outpatient treatment. (Strong recommendation; level I evidence).” In addition to objective data, the IDSA also recommends supplementing with subjective factors, including the ability to safely and reliably take oral medications and the availability of outpatient support resources (Strong recommendation; level II evidence). (Mandell, 2007).
Bottom Line: Based on the validated prognostic scoring systems and recommendations from the IDSA, a subgroup of low-risk patients with community acquired pneumonia can safely be managed as outpatients. Their prognosis can be reliably predicted by 3 scoring systems, where the PSI performs slightly better but is more complex to apply than the CURB and CURB-65.
- Which patients with influenza do you treat with oseltamivir?
Neuraminidase inhibitors, specifically oseltamivir, have been increasingly used in the last 5 years for the treatment of patients with symptoms of influenza. In fact, between May and December of 2009, around 18.3 million prescriptions were written for the drug in seven countries (Australia, Canada, France, Germany, Japan, UK and USA) (Muthuri 2014). Additionally, hundreds of millions of doses have been stockpiled in various countries as a safe guard against pandemic influenza and the World Health Organization (WHO) lists oseltamivir as an essential drug. Physicians have been encouraged to prescribe osletamivir to patients by drug companies, professional society recommendations, hospitals and by patient pressure. Despite widespread use, the evidence of benefit for oseltamivir has never sat on firm evidence-based grounds. Much of this stems from the reluctance of the pharmaceutical giant Roche to release all relevant study data on the drug. This changed in 2013 when all of the data was made available for analysis.
Before we delve into the recently released data surrounding oseltamivir, let’s look at the prior recommendations and the basis for these recommendations. In 2009, the BMJ published a review of a number of observational studies looking at the effect of oseltamivir in the treatment of influenza (Freemantle 2009). This publication looked at randomized controlled studies provided by Roche pharmaceutical at that time. The limited available evidence supported the role of oseltamivir in reducing the rate of post-influenza pneumonia in otherwise healthy adults. There was no evidence of a mortality benefit and limited safety data (Freemantle 2014). Additionally, the available data supported the idea of using oseltamivir for chemoprophylaxis in patients who were at risk of exposure. A Cochrane group review in 2010 echoed these results but also stated that there was extensive bias present in the available studies and without a full disclosure of all the research, no strong recommendation could be made (Jefferson 2014). In spite of the limited evidence, broad recommendations were made including treatment of patients with multiple comorbidities, pregnant patients, those with immunocompromise and chemoprophylaxis for close contacts (Harper 2009).
In 2013, Roche pharmaceuticals released all of the study data. The Cochrane Respiratory group subsequently published an updated systematic review of all of the randomized controlled trials (Jefferson 2014) as well as a summary statement in the BMJ (Jefferson 2014). A number of statements from the prior review stand: there are minimal studies on efficacy and safety in pregnant patients, and no mortality benefit was seen. They did not find a reduction in post-influenza pneumonia and posited that this prior finding was likely due to publication bias. The below table summarizes the major outcomes of the 2014 Cochrane systematic review:
| Outcome Measure | Finding |
| Alleviation of Symptoms | Shortened by 16.8 hrs with oseltamivir |
| Admission to Hospital | No Difference |
| Reduction in Confirmed Pneumonia | No Difference |
| Other Complications | No Difference |
| Transmission in Prophylaxis Group | No Reduction |
Additionally, the group reported on a number of common side effects:
| Side Effect | Results |
| Nausea | Increased (NNH 28) |
| Vomiting | Increased (NNH 22) |
| Psychiatric Events | Increased (NNH 94) |
| Headache | Increased (NNH 32) |
Overall, we see a mild shortening in the duration of symptoms with no reduction in admission, confirmed, post-influenza pneumonia or other complications. The same findings were seen in pediatric patients as well. There is minimal evidence in regards to efficacy or safety in pregnancy as pregnancy was an exclusion criteria in most of the studies. Side effects were common. Also, chemoprophylaxis did not reduce transmission of the disease. These results call into question the utility of oseltamivir for the treatment of influenza in any patient.
In June 2014, the PRIDE Consortium Investigators published a study challenging the Cochrane group findings (Muthuri 2014). In this large observational cohort (n=29,234 patients) Murthi et al found an association with decreased mortality (adjusted OR = 0.81) and an additional benefit to early (< 2 days) treatment versus later treatment (adjusted OR = 0.48). This study, however, has major flaws and biases that question the validity of their conclusions. Only 19% of centers that were contacted agreed to contribute data to the Consortium. Thus there is a high potential for bias. Additionally, the researchers do not assess the quality of the studies included in their meta-analysis (Antes 2014). Regardless, observational data should not be used to trump the RCT data included in the Cochrane review.
Bottom Line: The best available evidence demonstrates that oseltamivir leads to a mild reduction in the duration of symptoms of influenza. There is no proven benefit for mortality, hospital admissions or confirmed influenza related complications including pneumonia. The frequency of side effects may outweigh the mild symptom reduction benefit of the drug. The results of the 2014 Cochrane meta-analysis should be used to update the current CDC recommendations.
4. Which adult patients getting worked up for a urinary tract infection do you send a urine culture on?
A Urinary tract infection (UTI) is a condition in which bacteriuria is present with evidence of host invasion (presence of dysuria, frequency, flank pain, or fever). The “gold standard” in defining significant bacteriuria is the detection of any microorganisms by suprapubic aspiration. Since this method is not typically employed, many sources utilize a definition of more than 105 cfu/ml on a midstream urine culture to indicate true infection.
The American College of Emergency Physicians has recently kicked off the “Choosing Wisely” campaign (ACEP 2013) in an attempt to limit unnecessary testing in the Emergency Department. Although urine culture was not one of the five tests that was addressed, it is one of the most commonly sent laboratory tests in the ED making it a potential place to curb costs. In 1997, UTIs accounted for one million ED visits in the United States (Foxman 2002). Numerous publications and practice guidelines have recommended against the use of routine urine cultures in uncomplicated UTIs. Despite this, a 1999 survey of 269 EM physicians showed that 24% of them would order a urine culture on a 30-year-old non-pregnant woman with an uncomplicated UTI with dysuria of recent onset (Wigton 1999).
Werman et al in 1986 tackled the question of the “Utility of Urine Cultures in the Emergency Department” (Werman 1986). They concluded that urine cultures should only be obtained in patients at high risk for pyelonephritis or bacteremia/urosepsis, as well as in those expected to have uncommon or resistant organisms. This paper cited studies showing that routine urine cultures in nonpregnant women with acute cystitis do not affect management. Morrow et al showed that treatment of women seen in the ED with suspected cystitis proceeded with little attention to urine culture results, despite the fact that cultures were obtained routinely (Morrow 1976). Winickoff et al demonstrated that patients who did not have follow-up urine cultures after a UTI had no greater risk for reinfection or complications than did patients in whom follow-up cultures were obtained (Winickoff 1981). Additionally, a positive culture does not necessarily indicate the absolute need for antibiotics. In a study of 53 women with culture-proven UTIs, no patient progressed to pyelonephritis or bacteremia in spite of the fact that all were treated with placebo (Mabeck 1972).
In 2011, Johnson et al. addressed the question “Do Urine Cultures for Urinary Tract Infections Decrease Follow-up Visits?” (Johnson 2011). This retrospective cohort study looked at 779 female patients age 18-65 diagnosed with a UTI or acute cystitis treated in a family medicine clinic (exclusion criteria: pregnancy, diabetes, UTI or antibiotic use in preceding 6 weeks, other medical condition making UTI complicated). The follow-up rate for patients without urine cultures was 8.4%, which showed no statistical difference between the follow-up rate of 8.7% for patients with urine cultures. Ordering a urine culture was not associated with a decreased rate of follow-up visits (adjusted OR 1.11 [CI 0.65-1.90]). Of all 447 urine cultures ordered, only 1 grew bacteria resistant to nitrofurantoin, a common antibiotic used in the ED for uncomplicated cystitis. A 2006 UK study found that 23 women required a urine culture to prevent one follow-up visit from resistance-based failure; thus, empiric treatment with no urine culture was recommended (McNulty 2006).
Bottom Line: Although there are no prospective randomized controlled trials looking specifically at ED patients with UTI symptoms, it is safe to say that a urine culture in healthy adult non-pregnant females with new onset urinary symptoms without concern for pyelonephritis or bacteremia is unlikely to change management or outcome.


Pingback: Low-Risk Chest Pain Controversy: The FOAMed Report - news from Allwebsolutions.net
Pingback: Low-Risk Chest Pain Controversy: The FOAMed Report | multinationaltelecommunications.com
Pingback: LITFL Review 171 - LITFL
Pingback: LITFL Review 171 • LITFL Medical Blog • FOAMed Review